An innovative simple home-use diagnosis solution for sleep apnea
Approximately 175 million Europeans have obstructive sleep apnoea, a chronic condition characterised by regular pauses in breathing during sleep and associated with an increased risk of mortality. This is according to a recent study by connected health company ResMed, who in earlier work put the total number of people with sleep apnoea worldwide at a staggering 936 million – almost ten times higher than that previously estimated by the World Health Organization.[1]
While there are no studies estimating an exact figure, we can imagine that the impact is significant in terms of quality of life and long-term health risks for those living with untreated sleep apnoea, as well as the toll on companies and Europe’s healthcare systems.[2]
In 2015, undiagnosed sleep apnoea cost the United States nearly $150bn (~€130bn), as a result of related lost productivity, motor vehicle accidents and workplace accidents, as well as healthcare costs and medications prescribed to treat conditions associated with these untreated apnoeas.1 Europe has threefold the amount of people living with sleep apnoea as the United States, and it’s reasonable to imagine that the impact is significantly higher as well.[3]
Over 30 million people are undiagnosed for sleep apnea in Europe. Polysomnography (PSG) is the gold-standard diagnostic technique for sleep apnea, but it involves an overnight stay in a hospital or sleep unit and is costly and labour-intensive.
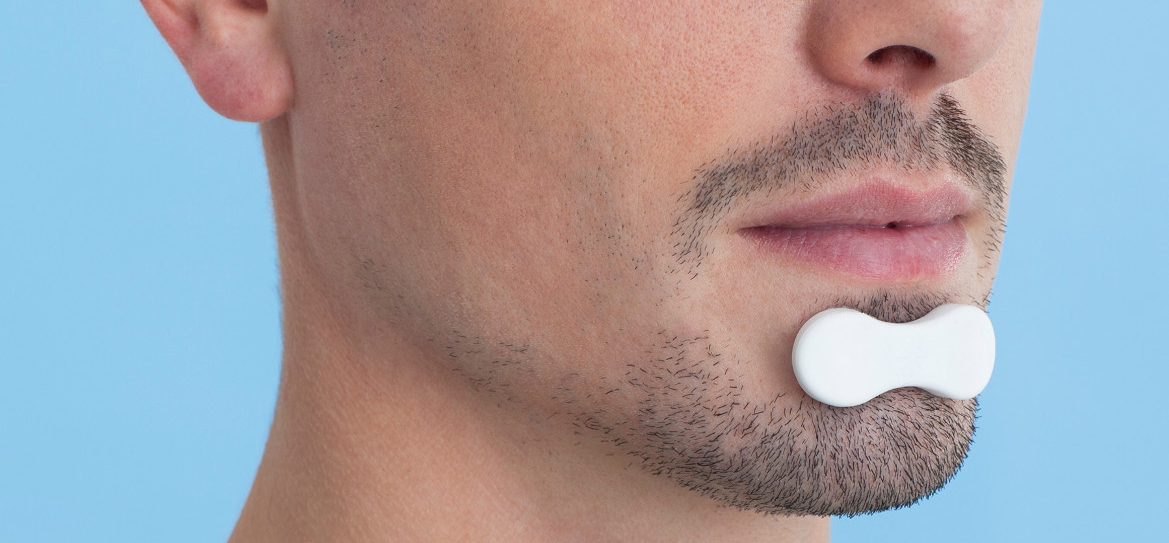
SENSAPNEA is an entrepreneurship project for the development of a simple solution for sleep apnea diagnosis and value-based care. The solution is easy for patients to use in their own homes and is cost-effective for both patients and physicians.
The proposed solution has three components:
- a chin sensor recording mandibular movements, which represent a reliable marker of respiratory efforts;
- a mobile app for patient-operated data recording and dashboard visualization;
- a cloud-based platform for patient data management
The innovative, simplified sleep apnea diagnostic promises better prevention and management of chronic diseases, at a reduced cost. It also empowers citizens to take charge of their own health with a digitally-enabled platform that involves joint-management with patients.
Thanks to an easy-to-use, wireless, 3-gram sensor, Sunrise allows anyone to clinically test its sleep. It brings gold-standard quality from hospital diagnosis directly to the patient’s home and it is the only certified medical device fully automating its analysis while demonstrating above 90% agreement with in-lab sleep studies. At a fraction of the cost.
Milestones
- 2019 – The consortium is created and the project begins
- 22 January 2020 – Scientific publication, Assessment of a mandibular movement monitoring with machine learning analysis for the diagnosis of obstructive sleep apnea: the study validates the effectiveness of the sensors in the device
- 12 November 2020 – The French High Authority for Health accepts to refund the SUNRISE scheme in the framework of the Innovation package
- 8 January 2021 – The start-up Sunrise is awarded with the “CES Innovation Awards Honoree”
- 1 March 2021 – Scientific publication, Clinical validation of a mandibular movement signal based system for the diagnosis of pediatric sleep apnea: A prospective study in 140 children undergoing in-lab polysomnography evaluates the accuracy of Sunrise for the diagnosis of OSA. The results show highly reliable results for moderate to severe OSA patients, that is, those clearly requiring treatment interventions.
- 17 March 2021 – Sunrise receives the IoT Awards of the Sleep solution of the year.
« Sunrise is the only certified medical device with fully automated analysis demonstrating agreement over 90% with in-lab sleep studies, at a fraction of the cost.”
Laurent, CEO of Sunrise
« The Fondation de l’Avenir, along with its partners, is particularly proud to support the Sensapnea project, a pioneer in the implementation of a solution contributing to the improvement of the diagnosis of sleep apnea, and to contribute, with regard to its social missions, to the outreach of medical research in Europe »
Marion LELOUVIER, Présidente du directoire de la Fondation de l’Avenir
“This product showcased innovative design and engineering features that scored highly across the evaluation criteria and joins an elite group of other products given this honor. Congratulations on this accomplishment!”
- A member of the Jury from the CES
“The medico-economic analysis carried out by PRECIS, AÉSIO Santé’s clinical research centre, shows that this device is as effective as it is inexpensive. It is therefore a solution that is in line not only with our high standards, but also and above all with our desire to support innovation for the benefit of all. “
Rémi BOUVIER, Directeur Général d’AÉSIO Santé
“Revolutionary on the approach with a lightweight sensor connectable to his phone. The results are received very quickly and I can send them to my doctor directly from the app!”
Michael, 42 years old
“Very user-friendly! I had to take a sleep test and I wasn’t thrilled when I saw the hospital devices with all those cables. Very easy to use application and you can do it at home.”
Mélanie, 28 years old
Partners involved in the project
- Université Grenoble Alpes: the project coordinator
- Sunrise: external partner
- Imperial College London: clinical research
- MADoPA: living lab
- Fondation de l’Avenir: cost analysis
- Beau Soleil de Montpellier, groupe AÉSIO Santé: participated in the economic evaluation of the solution
- Docaposte: data storage
- Probayes: Artificial intelligence
Sources
[1] https://www.healtheuropa.eu/sleep-apnoea/88470/