Diabeloop for Teenagers
D4Teens seeks to adapt an “artificial pancreas” to better meet the needs of teenagers with Type 1 Diabetes. The DBLG1TM system, which is already being manufactured for use by diabetics, will be made more compatible for this key cohort of patients by addressing the physical and psychological needs of teenagers.
Type 1 Diabetes is an auto-immune chronic illness with half of the subjects diagnosed before the age of 20. DBLG1TM operates in a closed feedback loop, requiring only marginal human intervention. It helps patients maintain normoglycemia (appropriate blood-glucose levels) using automatic insulin dosage. The D4Teens project adapts the DBLG1TM for the specific medical and psychosocial needs and requirements of teenagers.
Team
Diabeloop SA will be the manufacturer of the medical device, while CERITD is the initiator of the artificial pancreas and has worked since 2011 with CEA & Diabeloop on the technology. Beyond these three French members, the Swedish RISE will focus on marketing for the Nordic countries, the German Profil GmbH will contribute on networking with stakeholders and the Belgian KUL will perform clinical trials.
The project
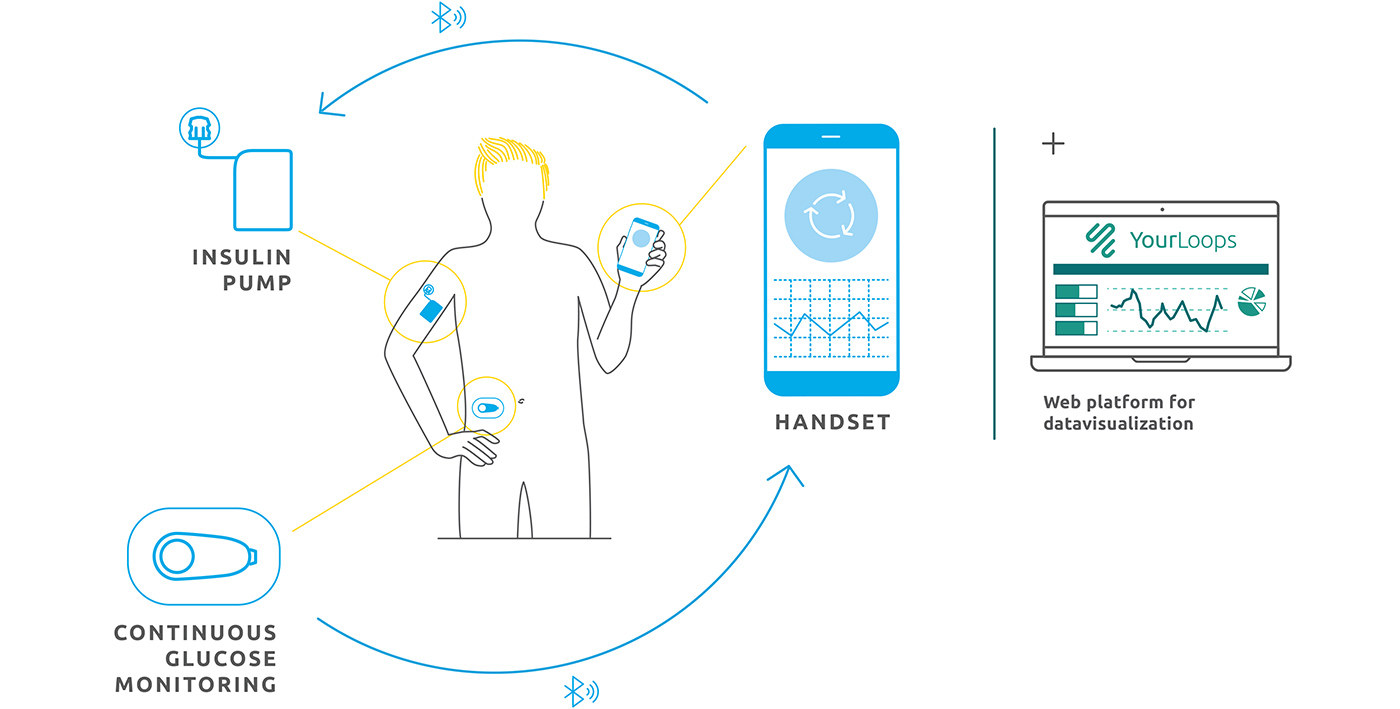
D4Teens adapts the DBLG1TM “artificial pancreas” for the specific needs and requirements of teenagers. The DBLG1TM system treats people with Type 1 Diabetes using three components connected in a closed feedback loop:
- A sensor monitors blood sugar (glycaemia).
- The sensor’s signals are transmitted by Bluetooth to a handset, where software runs complex algorithms, simulating human physiology to compute the right insulin dose.
- The dosage is administered by an insulin pump.
The system has proven effective, but it could better target the large cohort of teens with Type 1 Diabetes. Teenagers with Type 1 Diabetes face specific challenges with their insulin therapy, including issues related to hormonal transitions, the search for autonomy and the social pressure that is typical for this time of life.
With EIT Health’s support and networking possibilities, D4Teens will develop an improved system designed to maintain teenagers with Type 1 Diabetes in normoglycemia as much as possible, in order to mitigate comorbid conditions of diabetes. The adapted DBLG1TM should improve the teen’s treatment compliance, to allow a better start in adulthood.
Impact
- Teenaged users will benefit from enhanced management of Type 1 Diabetes.
- The healthcare system will benefit because young people develop life-long habits of better management of their Type 1 Diabetes.
- The commercial success of the device will generate high-quality jobs in the EU.
- Developments in a DBLG1TM version for teens will inform innovation in the “mainline” adult version and respond to the needs of others, including those with gestational or brittle diabetes.
Why this is an EIT Health project
This project is in keeping with the EIT Health Focus area of Bringing Care Home because the DBLG1TM system makes it easy for young people with Type 1 Diabetes to handle their care appropriately. It is also in line with the Focus Area of Care Pathways, because it seeks to improve the lifetime health outcomes of young people with diabetes.
External partner
- Diabeloop SA
An unmet need and a real improvement for people with diabetes. The DBLG1TM solutions will meet great demand and interest. It has the potential to simplify the lives of many people with diabetes.
Marlène Mehl International Development Lead
Members

CLC/InnoStars: France
Partner classification: Research, Tech Transfer, Clusters, Other NGOs
The CEA is a public organisation,leader in research, development and innovation, active in 4 main areas: low-carbon energies, defense and security, information technologies and technologies for health, promoting the transfer from research to industry.
Key Activities in Business Creation
Incubation, Technology Transfer


CLC/InnoStars: France
Partner classification: Business
Partner type: Network Partner
A non-profit association, (governed by the law of 1 July 1901) recognized as being of “general utility”, founded in 2006. An innovative clinical research centre in the field of diabetes providing patient care (as an outpatient structure of the Hospital), therapeutic education and performing clinical translational and applied research at the service of diabetic patients
Key Activities in Research and Developement
Biomedical engineering, Clinical research
Key Activities in Corporate Innovation
Med Tech, ICT, Diagnostics
Key Activities in Social Innovation
Healthcare provision
Key Activities in Business Creation
Technology Transfer, Testing & Validation
Key Activities in Education
Technical faculties, Medical faculties, Healthcare professional education/training


Partner classification: Education, Research
KU Leuven (together with University Hospitals Leuven) is a research-intensive, internationally oriented university that carries out excellence-driven research in health and care and is dedicated to build bridges between science, society and industry.
